Fluorescence is a phenomenon that causes a mineral to "glow" in the
within the visible spectrum when exposed to ultraviolet light. Minerals that exhibit
fluorescence are known as "fluorescent minerals". Fluorescent minerals contain
particles in their structure known as activators, which respond to ultraviolet
light by giving off a visible glow. Ultraviolet light is a form of electromagnetic radiation invisible to the
human eye. This light is given off by the sun and by common fluorescent lamps,
which also give off considerable white
light (visible light), preventing the fluorescence from being seen. The ultraviolet
reaction is only visible with a special fluorescent lamp with a filter that blocks white light
but allows ultraviolet light to pass through. This lamp is known as an ultraviolet
fluorescent lamp, or UV lamp.
Obviously, the reaction will only be visible in a dark area, where the presence of white
light is minimal.
There are two classified ultraviolet wavelengths: longwave and shortwave. Some minerals fluoresce
the same color in both wavelengths, others fluoresce in only one wavelength, and yet
others fluoresce different colors in different wavelengths. Some UV lamps have two
separate filters: one for longwave and the other for shortwave. Many more minerals
fluoresce in shortwave than in longwave; only a small amount fluoresce
in longwave. Longwave fluorescent lamps are fairly inexpensive, whereas shortwave lamps
are more costly. When this guide labels a mineral as fluorescent without specifying longwave
or shortwave, shortwave fluorescence is implied.
Fluorescence is not always reliable method for mineral
identification, since certain minerals of the same species may fluoresce different colors
from different localities, but it can still be an indicator. Once a mineral has been identified, its fluorescent color
is
sometimes used to identify its place of origin. Similarly, if an unidentified mineral
from a certain locality glows a specific color, it can often be identified by the color of
its fluorescence.
Color and intensity of the fluorescence varies among
specimens of a particular mineral. However, specimens from the same locality virtually
always fluoresce the same color. Calcite may fluoresce red, orange, yellow, white,
and green, but it will always fluoresce red at Franklin, New Jersey, and bluish-white at Terlingua, Texas.
When a fluorescent lamp is lit, never look directly at the light
source, as it can permanently damage the eyes. In addition, skin should not be exposed to
the light source for extended periods, as it can cause sunburns and long term skin
problems.
Below is a fluorescent Willemite and Calcite specimen from the Sterling Hill Mine in Ogdensburg, in the Franklin
District, New Jersey. The Willemite fluoresces green, and the Calcite red.
Specimen under UV Illumination:
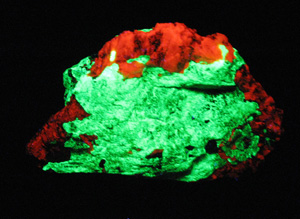
Specimen under standard Illumination:

(Collection and photos: Hershel Friedman)
Phosphorescence is a phenomenon exhibited in several fluorescent minerals where the
mineral continues to glow even after the UV light source has been removed. The glow slowly
fades, and after several seconds (or minutes in a few cases) is no longer visible to the
eye. Only few minerals phosphoresce; phosphorescence is only noted as interesting mineral
property, rarely as a diagnostic identification property. Certain mineral specimens from
specific localities all phosphoresce. Calcite specimens from Terlingua, Texas fluoresce
bluish-white with a strong phosphorescence.